|
Animals eat the plants so they too have carbon-14 in their tissues. but the carbon-14 decays away, at a constant rate, making carbon-14 a ticking atomic clock. 14 N (n,p) 14 C 14 C can also be produced by the following reaction: 13 C(d,p) 14 C d: deuterium or 2 H the production rate is 2.4 0.2 atoms (cm 2 sec)-1 Radioactive decay 14 C decays by b-decay with a maximum. The length of time it will take for half the amount of carbon-14 to decay is known as its half-life. 
Plants take in carbon-14 through the process of photosynthesis. Natural 14 C production 14 C is mainly produced by interaction of cosmic ray derived secondary neutrons with 14 N in the atmosphere. Solution One mole of carbon has a mass of 12.0 g, since it is nearly pure 12 C 12 C. The amount of carbon-14 in the atomosphere is, on an average, relatively constant. A sample of radioactive isotope A and a sample of radioactive isotope B decay as shown in the graph below. While an organism is living, it takes in carbon from the atmosphere and the ratio of carbon-14 to the stable isotope carbon-12 in the organism is constant. The decay constant is determined from the known half-life of 14 C 14 C (available from Figure 10.4). A species half-life is the time is takes for that species to decay. The 14C/ 12C ratio in living organisms is 1. Carbon-14 is a radioisotope formed in our atmosphere by the bombardment of nitrogen-14 by cosmic rays. Radioactive dating is a process by which the approximate age of an object is determined through the use of certain radioactive nuclides.For example, carbon-14 has a half-life of 5,730 years and is used to measure the age of organic material. The value of k for carbon-14 is 1.21 x 10-4 year-1, which comes from the half-life formula. 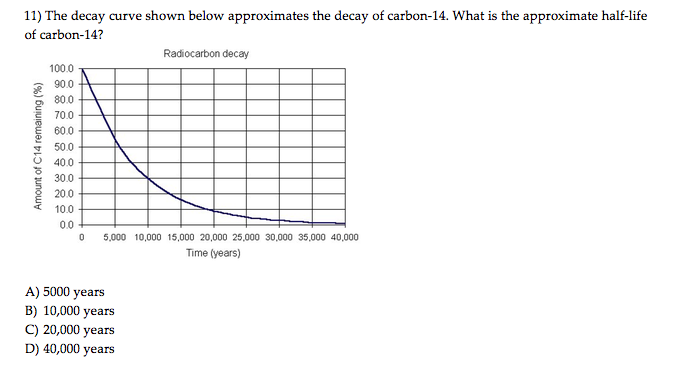
The half-life for this reaction is 5700 ± 30 yr. Carbon-14, 14 C General Symbol: 14 C: Names: carbon-14, 14C, C-14, radiocarbon: Protons (Z) 6: Neutrons (N) 8: Nuclide data Natural abundance: 1 part per trillion / Half-life (t 1/2) 5730 30 years: Isotope mass: 14.0032420 Da: Spin: 0+ Decay modes Decay mode: Decay energy : Beta: 0. 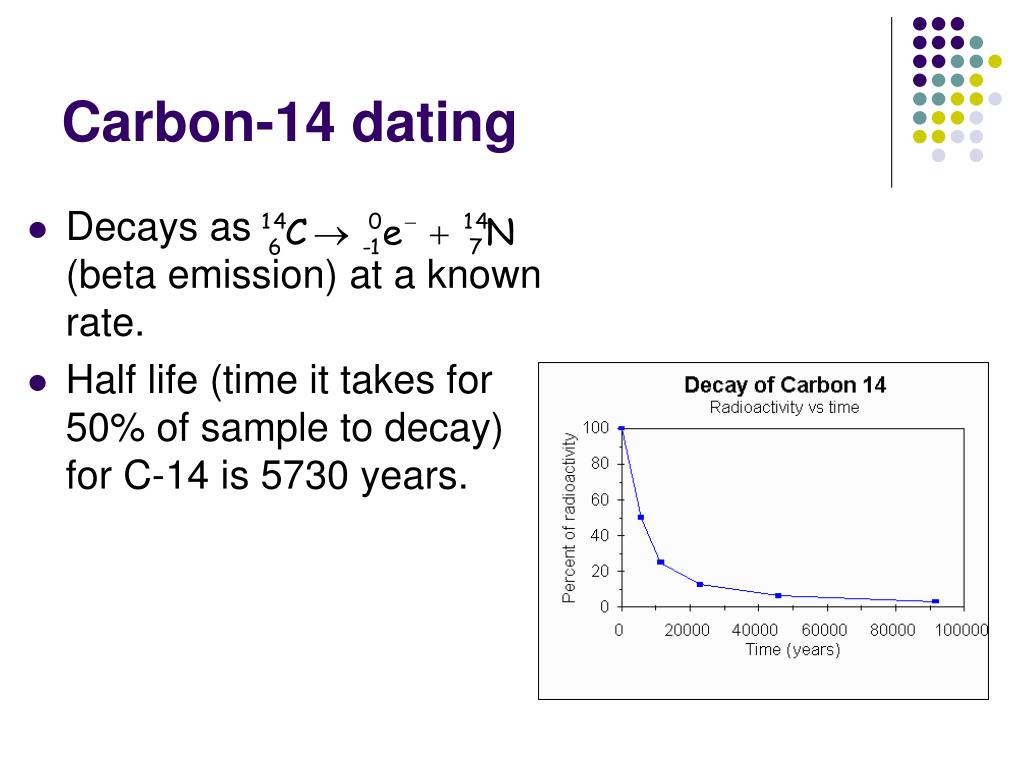
A plot of the specific activity of 14C versus age for a number of archaeological samples shows an inverse linear relationship between 14C content (a log scale) and age (a linear scale).\]
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
